About UCSF CIRP
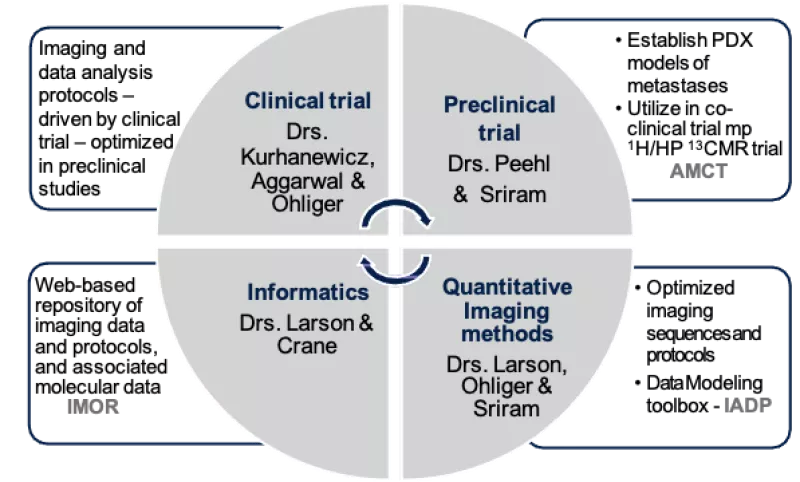
Project Aims
While the quantitative HP 13C MRI acquisition and data analyses tools developed in this U24 grant are focused on assessing chemotherapy of advanced prostate cancer, they will generally be applicable to other treatments and diseases, benefitting the community at large.
Aim 1: Optimize and validate
appropriate preclinical HP 13C MRI protocols and analyses that are
consistent withthose utilized in patient studies using realistic PDX models of metastatic SCNC.
- Evaluate the optimal radiofrequency (RF) coil set-ups for imaging SCNC tumors in liver and bone.
- Optimize a dynamic fast HP 13C echoplanar imaging MRI acquisition to acquire metabolic data of high spatial and temporal resolution with sufficient signal-to-noise ratio.
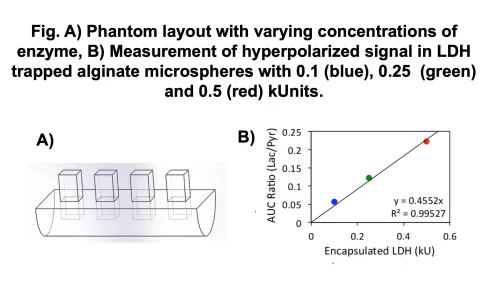
- Develop metabolic phantoms to test reproducibility and robustness of kPL measurements using optimized RF coils and pulse sequences.
- Evaluate the inputless model to calculate kPL, and validate using metabolic phantoms and preclinical murine models.
Aim 2: Implement the optimized preclinical dynamic HP 13C MRI protocols and data modeling approaches developed in Aim 1 to assess whether kPL can be used as a biomarker of therapeutic response or resistance.
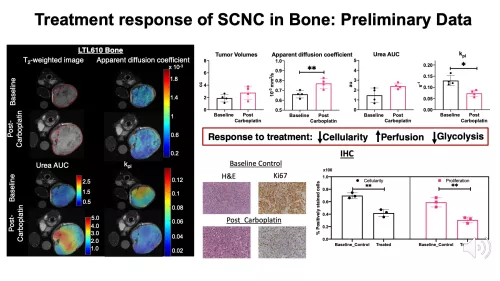
- Assess the changes in kPL of SCNC PDX tumors in liver and bone to platinum-based chemotherapy as a marker of response or resistance. The preclinical HP 13C MRI findings will be used to inform on HP 13C MRI studies of patients with SCNC of liver and bone metastases before and after platinum-based chemotherapy.
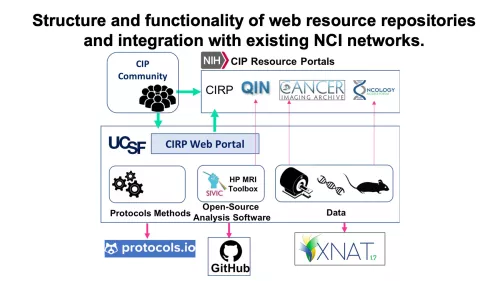
Aim 3: Establish a web-based repository of co-clinical imaging data and protocols, associated molecular and pathological characterization of PDX models, processed data and processing pipelines, as well as the analytical tools developed.
- Characterize the transcriptomic and metabolic profiles of the PDX lines in the two different metastatic sites and supplement a comprehensive information resource on Oncology Models Forum.
- Institute an online portal to establish a repository of data, protocols and tools for image acquisition and quantitative processing.
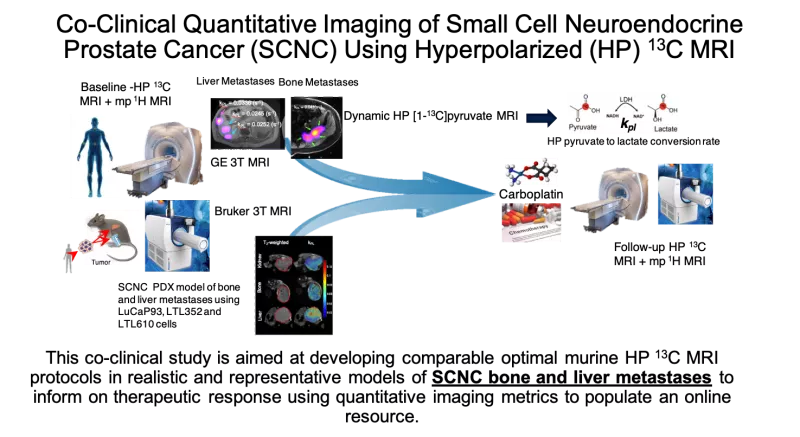
This co-clinical project is based on the goal of assessing therapeutic response of small cell neuroendocrine prostate cancer (SCNC) using quantitative hyperpolarized (HP) 13C-pyruvate magnetic resonance imaging (MRI). The murine imaging study will be conducted in parallel to a clinical trial supported by NCI R01 CA215694, aiming to assess response of SCNC to carboplatin in men with metastatic prostate cancer (PCa), led by Drs. John Kurhanewicz and Rahul Aggarwal. Although next-generation androgen receptor signaling inhibitors have significantly improved outcomes in men with metastatic castration-resistant prostate cancer (mCRPC), these agents frequently induce SCNC, a lethal subtype of mCRPC, with median survival of <9 months. While platinum based chemotherapy is the standard of care for treatment-induced SCNC (t-SCNC), newer therapies targeting the underlying molecular events of transdifferentiation and progression of mCRPC to t-SCNC are impeded by lack of predictive biomarkers. The selection of the most appropriate treatment is hindered by the fact that neither blood tests (such as serum neuroendocrine markers) or current imaging modalities such as multiparametric 1H MRI or positron emission tomography (PET) (e.g., somatostatin receptor-based or 18F-fluorodeoxyglucose) can reliably identify therapeutic efficacy in these metastatic tumors which are also often not amenable to biopsy. HP 13C-pyruvate MRI is a new, safe, non-radioactive, quantitative stable-isotope imaging approach that has the potential to enable paradigm-shifting, real-time monitoring of metabolic response to treatment in vivo. Preliminary data suggest that a rapid (~5 minute) HP MRI quantitative measurement of the rate of conversion of pyruvate to lactate, kPL, can reflect changes in metabolic reprogramming that occur upon early response to conventional treatment (carboplatin) of t-SCNC. This proposal is timely since standardized data acquisition and data analysis approaches as well as quantitative imaging metrics for HP 13C MRI need to be established for ongoing clinical trials of cancer detection and therapeutic response. The study design of this project fits the goals of the NCI Oncology Co-Clinical Imaging Research Resources Program (CIRP) and aims to use appropriate patient-derived xenograft (PDX) models that reflect the genetic, metabolic and micro-environmental heterogeneity of SCNC. Quantitative HP 13C MRI acquisition and data analysis methods will be optimized and applied to study the response of these PDX models of SCNC to chemotherapy, paralleling the funded study in patients. This co-clinical study is aimed at developing optimal HP 13C MRI protocols in mice that mimic parallel clinical protocols, and modeling the dynamic HP 13C MRI data to determine changes in kPL, in order to predict therapeutic response/resistance of realistic PDX models of SCNC bone and liver metastases. The deliverables of this research will include an online resource of quantitative HP 13C MRI imaging protocols, data analyses and modeling tools as well as correlative biology data for wider dissemination and validation by the scientific community.
UCSF CIRP Team

John Kurhanewicz, PhD
Professor
Preclinical / Clinical Trials & Quantitative Imaging Methods
[email protected]

Renuka Sriram, PhD
Assistant Professor
Preclinical Trials, Quantitative Imaging Methods & Informatics
[email protected]

Michael Ohliger, MD, PhD
Associate Professor
Abdominal & Liver Radiologist
Clinical Trials
[email protected]

Robert Bok, MD, PhD
Professor
Preclinical & Clinical Trials
[email protected]

Rosie Nolley
Specialist
Histology
[email protected]

Avantika Sinha
Assistant Specialist
Preclinical Trials, Quantitative Imaging Methods & Informatics
[email protected]

Ivina Mali
Postdoctoral Scholar
Preclinical Trials
[email protected]

Lynn Delos Santos
Pathology/Molecular Biology
Preclinical Trials
[email protected]

Peder Larson, PhD
Professor
Quantitative Imaging Methods & Informatics
[email protected]

Jason Crane, PhD
Director, Informatics
& Scientific Software
Informatics
[email protected]

Rahul Aggarwal, MD
Associate Professor
Clinical Trials
[email protected]

Donna Peehl, PhD
Professor
Preclinical Trials
[email protected]

Shubhangi Agarwal, PhD
Postdoctoral Scholar
Preclinical Trials, Quantitative Imaging Methods & Informatics
[email protected]

Jennifer Lewis, PhD
Postdoctoral Scholar
Probe Formulation Optimization & Preclinical Trials
[email protected]

Jenna Bernard, PhD
HP Data Scientist
& Communications Manager
Website & Digital Resources
[email protected]
Facilities
The findings and tools developed within this CIRP are supported directly by the Surbeck Laboratory for Advanced Imaging and the Preclinical MR Imaging & Spectroscopy Lab. The Surbeck lab (housing the clinical GE 3T and 7T MRI along with two GE SPINlab DNP polarizers) and the preclinical MRI facility at the Mission Bay campus are 600 yards apart and are part of the hyperpolarized magnetic resonance technology resource center (HMTRC, an NIH-funded P41 EB013598 center). The GE 3T clinical scanner is equipped with a multitude of 1H and 13C coils that have been tested and validated for HP 13C MRI trials at UCSF. The preclinical MRI facility is also equipped with a Bruker cryogen-free 3T system along with two Hypersense (Oxford Instruments) polarizers, providing an ideal set-up for a co-clinical imaging study. We are also equipped with a state-of-the-art radio-frequency (RF) coil laboratory. Additionally, in the same building we have a high resolution 2D printer and machine shop critical to the development and testing of quantitative imaging phantoms.
Surbeck Laboratory for Advanced Imaging
The California Institute for Quantitative Biomedical Research (QB3) houses the Surbeck Laboratory for Advanced Imaging which includes a 7T GE 950 whole body scanner, a new 750 3T research scanner, microscopy, computational and other facilities. The Surbeck Laboratory also has facilities for managing human subjects including offices, a changing room, preparation room, and restroom.
Clinical Equipment:
3.35T GE SPINlab Polarizer
• Polarized sample temperature: ≥ 0.8K
• Polarization transfer source: 94GHz (up to 100mW)
• Polarization sample volume: 60ul to 1.5ml
• Polarization sample quantity: up to 4 sample vials simultaneous
• Daily sample quantity: up to 12 samples/regeneration cycle
• Polarization agent: Trityl radical, typical
• Dissolution volume: 15ml to 50ml, typically water based
• Helium consumption/dissolution: N.A.
• Sample polarization time: typically 15 mins to 2.5 hours
• Sample dissolution time: > 10 secs
• Sample 13C S/N enhancement: up to 10,000 fold
3T GE 750 Whole Body MR Scanner
• Discovery MR750 3.0T with 60cm bore
• 18 superconducting High Order Shims, including X, Y, Z, XY, XZ, YZ Z2, X2-Y2
• Optical RF technology with 165dB dynamic range. Scalability up to 136 coil connections
• Technology enables multi-channel broadband receive for multi-nuclear studies
• Processing power to reconstruct 32-ch of high-resolution 3D volumetric data. Vector Reconstruction Engine (VRE)
• Extreme Gradient Driver (XGD)
• Supports 50mT/m peak amplitude at a slew-rate of 200mT/m/s on all 3-axis
• RF amplifier: for body coil, water cooled, solid-state 35 kW RF amplifier. Supports a 100W continuous-wave output mode for proton-decoupling & spin-labeling research.
• Multichannel spectroscopy with 8-kW broadband amplifier and 8 ch receiver converter.
QTAR Coil

Preclinical MR Imaging & Spectroscopy Lab
The Preclinical MR Imaging and Spectroscopy Laboratory is located in Genentech hall, the building adjacent to the Quantitative Bioscience Institute (QBI), on the UCSF Mission Bay Campus. This state-of-the-art Core facility offers MRI and MRS capabilities which are uniquely integrated with DNP polarizers for conducting hyperpolarized 13C MR experiments.
Preclinical Equipment:
HyperSense DNP Polarizer
• Field strength: 3.35T
• Microwave source: Integrated 94GHz with 0.5Hz sweep, (user selectable) up to 100mW
• Sample volume: 10-200ul
• Dissolution volume: 3.5-5ml
• Dissolution solvent: water
• Temperature: (selectable) 1.3°K
• Nuclei: 13C, 15N, 31P, 29Si and other spin ½ nuclei
• Polarization duration: 15min – 6hrs
• Polarization agent: 2-5mg ‘Trityl radical’ per sample
• Helium consumption: 2L per dissolution
Polarize SpinAligner
Bruker BioSpec® 3T Cryogen Free Pre-clinical MRI
• A low-cost, cryogen-free superconducting magnet (18 cm bore), small footprint, 3 Tesla multinuclear MRI system optimize for murine and rat imaging
• Excellent field homogeneity (0.1ppm, over a 5 cm FOV) with high order room temperature shims (x,y,z, z2, xz, yz, x2-y2, 2xy, z3) and strong gradients (960 mT/m, 3,550 T/m/s)
• System is equipped with 1H and 1H/13C body & head coils for rats/mice
• Flexible MRI operating environment allows for both the development of advanced MRI imaging protocols and provide a more push button operation mode for less experienced users
Other CIRPS & Useful Links
Protocols and tools for image acquisition and quantitative processing can be found on this website as well as several other websites. Collected below are a list of links to other CIRP sites as well as useful links for web resources.
Co-Clinical Imaging Research
Resources Program (CIRP) Sites
NCI Co-Clinical Imaging Research Resource Program (CIRP) promotes development of quantitative imaging resources for therapeutic or prevention co-clinical trials that study both patients and human-in-mouse models.
For a comprehensive list of other CIRP Sites please visit the NCI Cancer Imaging Program (CIP) CIRP Website.
National Cancer Institute CIP Resource Portals
Oncology Models Forum (NCI OMF): For annotated data sets, analysis tools and more.
Quantitative Imaging Network (QIN): For more resources on the development and validation of data acquisition, analysis methods, and tools to tailor treatment to individual patients and to predict or monitor the response to drug or radiation therapy.
The Cancer Imaging Archive (TCIA): For publicly shared cancer imaging data. TCIA collections include radiology and pathology images, clinical and clinical trial data, image derived annotations and quantitative features and a growing collection of related ‘omics data both from clinical and pre-clinical studies.
NCI CIRP Hub: This public wiki site has 1) CIRP Network information 2) CIR Resources 3) Researcher Resources. This site also serves as a space for the network interactions, and the page for CIRP Members requires to login in.
The Hyperpolarized MRI Technology
Resource Center (HMTRC)
The Hyperpolarized MRI Technology Resource Center (HMTRC) is a Biomedical Resource Technology Center funded by the National Institute of Biomedical Imaging and Bioengineering (NIBIB NIH P41EB013598) and focuses on the development and dissemination of new advances in dissolution DNP techniques and instrumentation, specialized data acquisition methodology, and analysis software for biomedical research.
This website is a great resource for HP C-13 related tools, protocols, and trianings that involve both preclinical and clinical research.
UCSF Department of Radiology
& Biomedical Imaging
The Department of Radiology and Biomedical Imaging at the University of California, San Francisco, is a leading health sciences center focused on serving patients, conducting research, and training the next generation of radiologists.
This website is a great portal for the latest information on Patient Care, Reserach, and Education that is occuring within the UCSF Department of Radiology and Biomedical Imaging.